
Using the initial values of the model for all solute species, 1/κ for the thylakoid membrane is calculated as 61.5 nm (see Supplementary Material). what: The current model presented in this work can serve as a basis for further extension into a more detailed theoretical model by which multiple variables involved in photosynthesis can be explored.who: Hui Lyu and Dušan Lazár from the Foshan University, China have published the research work: Analyzing the effect of ion binding to the membrane-surface on regulating the light-induced transthylakoid electric potential (ΔΨm), in the Journal: (JOURNAL).The volume of the given matter is more when the thermal interactions are high. The volume of the given matter is less when the intermolecular forces are high. Thermal forces are weak in solid state and high in the gaseous phase. Intermolecular forces are weak in the gaseous phase and strongest in solid state. Thermal energy is directly dependent/proportional to temperature. They do not have any effect due to the temperature, but the boiling point of the substance is directly proportional to the strength of the forces. Greater thermal interactions will not allow the substance to remain in the solid state. Greater intermolecular forces lead to the substance being in the solid state. These forces are due to the motion of particles. These forces are due to the dipole of one or both the given molecules. The total measure of the sum of the kinetic energy of all the atoms and molecules is called thermal energy. The forces of attraction and repulsion between interacting atoms and molecules are called intermolecular forces. The difference between Intermolecular Forces and Thermal Interactions are as follows: The partially positive of the polar molecule attracts the partially negative part of another molecule.įor example, HCl molecules have dipole-dipole interactions. Their permanent dipoles due to varying electronegativities of the atoms are associated with a covalent bond. They have dipole-dipole interactions as attractive forces.  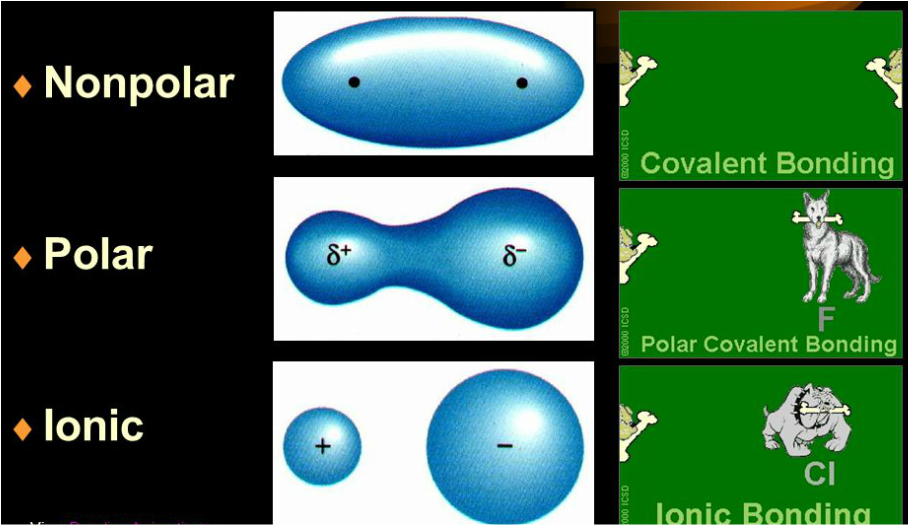
Polar Molecules are those molecules that have a net dipole because of opposing charges on either end. It is the weakest force out of all the intermolecular forces. London forces operate for a short distance (~500 pm) and their magnitude depends largely on the polarisability of the particle. In the final part, we can observe how Atom A has more electron density on the left-hand side. Next, we can see Atom A with an instantaneous dipole with more electron density on the right-hand side, while Atom B is with an induced dipole. ISRO CS Syllabus for Scientist/Engineer ExamĪs we can see in the image above, we first consider two atoms A and B that have no dipole moment.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.DevOps Engineering - Planning to Production. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
